Yeast Tetrad Dissection
What is yeast tetrad dissection?
Tetrad dissection has been a vital tool in yeast genetics since 1937 when Winge & Laustsen first teased apart the four spores of a tetrad and isolated the individual cells. Scientists used to do this manually with years of dexterity, skill and hard graft. Fortunately for modern researchers, we now have specially designed micromanipulation microscopes to make tetrad analysis easier. But it pays to understand the theory before mastering either technique.
Sporulation in yeast
Yeast cells don’t have male/female. Instead one MATa and one MATα strain fuse together, each providing one set of chromosomes to the diploid daughter cell in a cross. When the diploid cell is placed onto low-nutrient media, sporulation is induced during which diploid yeast undergo meiosis to create four haploid spores encased in the ascus membrane.
I.e. The resulting spores each contain half the usual amount of DNA (haploid). Yeast cells also readily undergo recombination when they divide, a feature that naturally generates genetic diversity within the population and is often exploited for genetic studies.
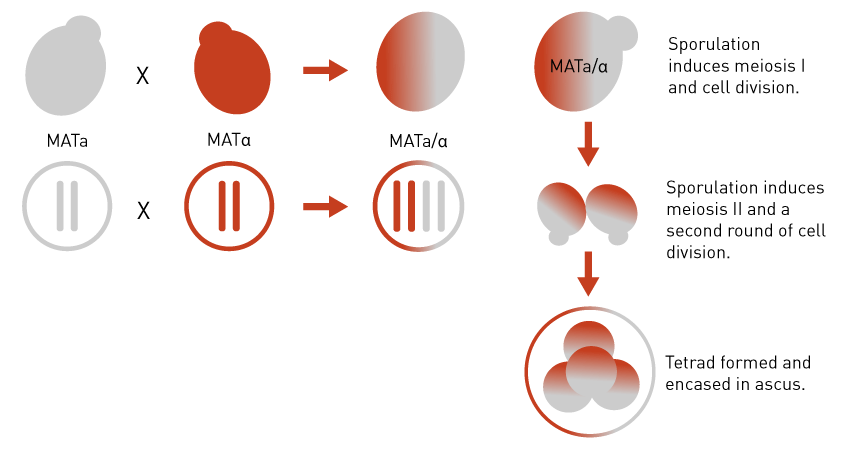
Figure 1: Diagrams illustrating the process of yeast mating and sporulation.
The Art of Tetrad Dissection for Beginners
When yeast cells divide through budding, the progeny are genetically identical to the parental cell. But when a scientist wants to study something like genetic linkage it is desirable to cross different strains of known genetic sequence and study the progeny. As previously explained, this mating process produces a tetrad. To determine the genetic make up of the spores, they must first be physically separated using tiny glass needles under a yeast tetrad dissection microscope.
Digestion
The first step is to break down the protective ascus membrane that surrounds the tetrad. An enzyme such as Zymolase is used in several parallel digestions across a few time points to obtain the best results. The tetrad should be sufficiently digested to dissolve the ascus but not so much that the spores separate. A well digested tetrad should collapse into a cruciform, shown in Figure 2. A protocol for tetrad digestion can be found here.
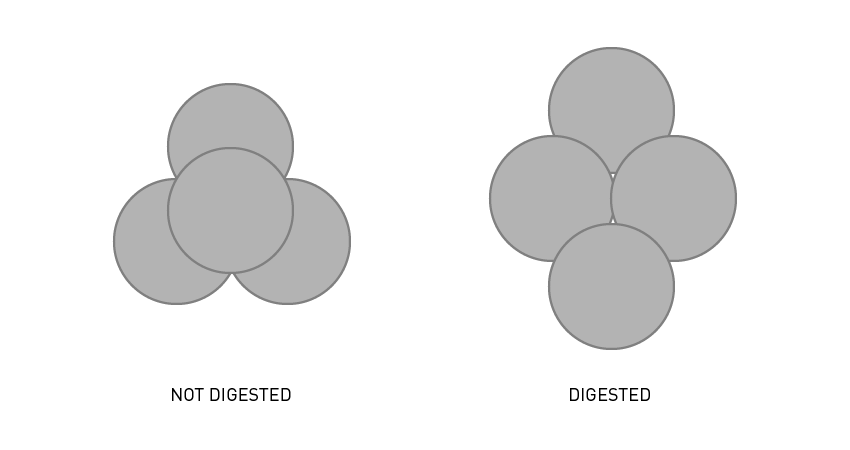
Figure 2: Diagrams indicating the appearance of a tetrad when viewed under a microscope. Both when undigested and when properly prepared, ready for tetrad dissection.
Dissection
A single line of digested material is streaked onto an agar plate or agar coated glass slide and inverted onto a microscope. From here yeast enthusiasts used to manually separate the tetrads using homemade needles whilst looking through the microscope. Very fiddly.
Fortunately for almost 40 years Singer Instruments have worked to make your life easier with our collection of dissection microscopes, complete with integrated micromanipulators. The late Carl Singer kicked off our obsession with yeast, with the Singer Instruments’ MSM system first launched in the 1950’s (Singer & Cooper, 2021). Since then, and with his son Harry now in charge, the MSM400 and the SporePlay dissection platforms continue to allow hundreds of scientists access to a semi-automated tetrad dissection solution. So you can focus on the fun stuff, like genetic linkage analysis.
Dissecting tetrads can be somewhat of a dark art. So to show you the ropes, here’s our very own Jan Singer. Jan honed his skills while teaching tetrad dissection to undergrad students on the Cold Spring Harbour Yeast Genetics and Genomics course. So you couldn’t have a better teacher!
What makes tetrad dissection so powerful?
Explore how scientists employ genetic linkage analysis to understand complex inheritance patterns in yeast and beyond.

Fiona Kemm MRes | Scientist
Fiona is a vital member of our Research team, rigorously testing our robots to ensure scientists don’t break them. With no prior robotics experience, she was the ideal guinea pig for our world-class user experience and support. Holding a BSc in Biochemistry and an MRes in Molecular Microbiology, Fiona brings extensive hands-on expertise she applies across departments, supporting both users and internal teams. From writing insightful web articles to specialising in SQWERTY, Fiona ensures our innovations perform flawlessly, helping customers focus on the creative and interpretive aspects of science that can’t be automated.